Basic Pathological Processes Seen With The Needle Examination
...
SPONTANEOUS ACTIVITY
Fibrillations And Positive Waves
When a muscle fiber is denervated, several pathological changes take place. The acetylcholine receptors spread all across the muscle fiber instead of being grouped in a well-defined geographical area, the end-plate. This spread may play a role in attracting new innervation to the denervated muscle fiber from adjacent nerve sprouts. The muscle fiber becomes much more sensitive to free acetylcholine released spontaneously from adjacent nerve fibers and is depolarized and repolarized spontaneously as these molecules reach it. Each single depolarization is electrically detected as a single muscle fiber action potential.
The Positive Sharp Wave
This wave represents a very sharp positive deflection off the baseline followed by a slower return and often a negative phase before returning to the baseline.

Positive sharp waves may reach up to 1 mv in amplitude and can last up to 50 msec. They discharge in a very rhythmic manner. Usually the rhythm starts and stops abruptly, and rarely does the individual rhythm vary.
The Fibrillation Potential
Of short duration (<3 msec) and low amplitude (<300 µv), fibrillation potentials occur in semirhythmical runs (<30/second), though occasionally the frequency is so slow it appears to be random.

With any new needle movement they may be activated again. Fibrillations are not seen immediately but develop two to three weeks after the neuron or axon has been damaged. Muscles closer to the neuraxis will develop them earlier than those in the distal part of the extremities. At times, the fibrillations have been reported as long as twenty years after denervation, though they are less frequently seen as time goes by and may be seen infrequently after three years.
As the muscle is reinnervated, both fibrillations and positive waves decrease in numbers and eventually disappear as reinnervation is successfully completed.
Fasciculations
A spontaneous discharge of an entire unit in a random fashion, the fasciculation looks like any motor unit seen but is distinguished by the irregular discharge pattern, and occurs spontaneously. Because many so-called fasciculations are nothing but units from poorly relaxed muscle, it is best to try and observe them clinically first. A genuine fasciculation will induce a noticeable needle movement when it occurs, whereas a poor relaxation causes little or no needle displacement. Fasciculation potentials can be monophasic or diphasic, looking like normal motor units, or highly polyphasic and complex, looking like neurogenic motor units. The former, the so-called benign fasciculations, are usually seen in normal persons having fatigue or muscle cramps and usually occur at 0.8 second intervals. The latter, the so-called malignant fasciculations, are seen most frequently in anterior horn cell disease, though they have been observed with chronic neuropathies, radiculopathies, and Creutzfeldt-Jakob disease and tend to have longer intervals between each other, usually in the vicinity of 3.5 seconds.
Complex Repetitive Discharges
Also known as high frequency discharges and bizarre repetitive potentials, these are long trains of rapidly firing potentials with abrupt onset and termination. These potentials, usually of low voltage and short duration, tend to group, firing at a frequency of 20-40/sec or higher.

They also tend to remain constant in size and frequency throughout the discharge. On occasion, the burst may only last l second during which the size of the potentials may change somewhat. These potentials are seen in a variety of myopathic and neuropathic conditions. Commonly seen in polymyositis and the early, active stages of Duchenne muscular dystrophy, they have also been described with myxedema. In neuropathic disease, they are seen in chronic root lesions, peripheral neuropathies, the motor neuron diseases and with nerve regeneration.
Myokimic Discharges
These are spontaneous bursts of rapidly firing potentials seen with clinical myokymia. These bursts recur at regular intervals of 2-10 per second and are unaffected by voluntary effort.
Neuromyotonia
These very high frequencies discharges occurring in long trains or bursts. Characteristically, their amplitude gradually decreases during the train or the burst, causing myotonic-like sound. They are typically seen in Isaac’s syndrome (continuous muscle fiber activity).
Myotonia
Probably the best-known sound in EMG to both the electromyographer and non-electromyographer is the so-called “dive bomber” sound produced by a myotonic discharge. This discharge can be triggered mechanically, electrically, or by needle insertion. Typically it consists of high frequency discharges that vary consistently in amplitude and frequency, waxing and waning continuously with firing frequencies ranging from 150/second down to 20/second and producing the dive bomber sound. When seen after insertion, these discharges are shaped like positive waves, whereas, after activation of the muscle, they look like spikes or fibrillation potentials and are called after-discharges. Insertional discharges and after-discharges may be so intense that any useful observation of the motor unit potentials between them may be impossible. They are seen in myotonic congenita, myotonic dystrophy, paramyotonia, and the hyperkalemic variety of the periodic paralyses. Unlike fibrillations and positive waves that decrease or disappear altogether with cold, myotonic discharges are greatly enhanced by lower temperatures.
VOLUNTARY ACTIVITY
The Neurogenic Motor Unit
When a muscle fiber is denervated, reinnervation can be accomplished in two ways, depending on the type of nerve injury.
In complete transection, after successful nerve repair, reinnervation takes place from regenerating new axons that reach the muscle fibers after they have traveled through the distal nerve stump. These axons reach the muscle at scattered time intervals. When they do, they attach themselves to muscle fibers that have been denervated for varying times and have belonged to different motor unit. Thus the motor units they form are small (100-200 µv) because of the atrophy of muscle fibers, of short duration (3 – 5 ms), and polyphasic (because of their lack of synchronization).
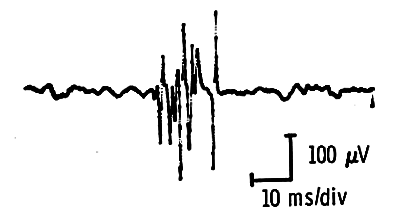
They are the so-called “nascent” potentials that are seen in the first two months after nerve injury and successful repair. Within four to six months the motor units become of longer duration, of higher amplitude, and are less polyphasic. At about eight months to a year they reach normal size for the muscle being examined. Often reinnervation by collateral sprouting (see below) takes place also, and chronic neurogenic units are seen along with these units.
In partial nerve injury, the type of lesion more commonly encountered in the EMG lab, reinnervation is accomplished by collateral sprouting with the denervated muscle fibers seeking new nerve sprouts from adjacent axons.
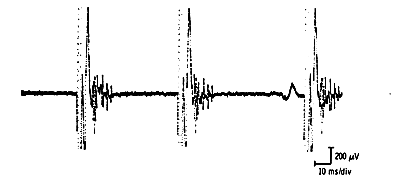
This reinnervation alters the motor unit in two ways: on the one hand, the motor unit now contains more muscle fibers; on the other hand, the newly acquired muscle fibers are asynchronous with those of the host unit and indeed also among themselves. The newly formed end-plate may not be stable in the beginning and many of them never reach maturity. Their respective muscle fibers either die or attract innervation from another source. This process of acquiring new muscle fibers and forming new end-plates begins in the first two months after nerve injury and results in a prolongation in the duration of the reinnervating motor unit duration and an increase in the number of its phases. The duration is prolonged simply because there are more fibers to depolarize, and the increase in the number of phases is due to the lack of synchronization between the host fibers and the newly acquired fibers.
Furthermore, since these newly acquired fibers have unstable and immature end-plates, neuromuscular transmission along them is erratic. This results in unstable components in these units. Long-duration polyphasic (by definition more than four phase) motor unit potentials with unstable components are indicative of the activity of the reinnervation process.
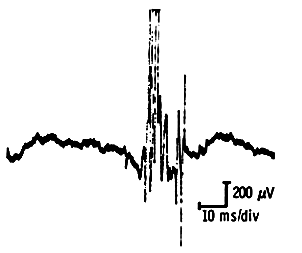
By six to eight weeks the fibers incorporated into the surviving motor units begin to take on the properties of these motor units and therefore become better synchronized with it. Conduction along the newly formed nerve sprouts becomes more stable. Therefore depolarization of the entire motor unit now takes less time, and the improved synchrony causes fewer irregularities in the depolarization-repolarization process, thuis reducing the number of phases. This chronic neurogenic motor unit, which is the end-stage of reinnervation, has a high amplitude and a long duration, and produces a typical thundery noise on the loud speaker. When its amplitude exceeds 10 mv, it is called a “giant potential”.
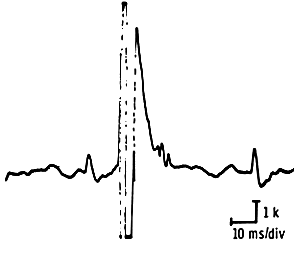
The Myopathic Lesion
In contrast to neurogenic lesions, nerve fibers in most myopathic lesions remain by and large intact while muscle fibers die or become diseased. Therefore, one of the typical early changes in myopathies is a reduced duration of the motor unit and a drop in its amplitude. The muscle fibers that survive are either still unaffected by the process or just beginning to be involved. These fibers will atrophy, divide, separate into small fragments, or split along their axes. Such changes result in a very erratic, unstable spread of the depolarizing current, causing considerable desynchronization in the motor units. Typically these motor units are of low amplitude, short duration, and have a high number of phases.
On the loud speaker, they have a typical scratchy metallic sound that can be best compared to the noise of hail falling on a tin roof. Another characteristic of these motor units is their recruitment in very large numbers at fairly low voluntary effort. Indeed, with only a moderate degree of contraction, one can see a full interference pattern. This results partially from the little effort that each of these reduced size motor units can deliver, thus requiring large numbers of them to deliver an adequate effort.
Changes Caused By Neuromuscular Transmission Defect
In diseases where the neuromuscular junctions is so impaired that transmission is either extremely erratic or does not take place, some muscle fibers affected by the process are “excluded” from the motor unit when their neuromuscular transmission fails. If a significant number of muscle fibers are thus “in” or “out” of the motor unit depending on neuromuscular transmission, moment-to-moment changes in the shape of the motor unit potential are seen as a result. These variations are present in the more severely affected muscles that, if accessible to nerve conduction studies, will show a decrement on repetitive stimulation.
